An affirmation of alternatives
11 Feb 2019 by Evoluted New Media
As concern over the reliability of commercially available antibodies continues to grow, we ask: when is an antibody not an antibody? When it is an affimer – Ryan Hannan and Robert Ford take us through the clinical-diagnostic successes of these non-antibody affinity reagents...
Diagnostic science has been revolutionised with the advent of monoclonal antibody technology. The ability to generate high volumes of specific binders to a wide range of targets has allowed the development of assays that analyse biomarkers to yield diagnostic and prognostic information on a range of diseases.
Antibodies form the basis of many of these assays, from pregnancy tests to the identification of infectious disease, and the diagnosis and monitoring of cancer patients. Earlier and easier diagnosis with such rapid, accurate assays has improved patient outcomes and reduced healthcare costs.
A mainstay of the antibody assay method has been plate-based enzyme-linked immunoassays (ELISAs), which are used across most fields of diagnostic medicine. This method of bioanalytical investigation provides quantitative information on the presence of specific biomarkers, and can be easily and rapidly performed by skilled workers in a laboratory environment. However, the need for trained laboratory workers to both perform and then interpret the ELISA assay results has led to an increased interest in simple point-of-care assays, which offer immediate results within the home, doctor’s office or the field, without the requirement for skilled staff.
Antibody alternatives As our knowledge and understanding of disease has increased, the number of validated biomarkers for different diseases has also expanded. Methods exploring the application of these disease biomarkers to clinical diagnostics have led to the development of biosensor and mass spectrometry-based assays, with increased specificity and sensitivity.
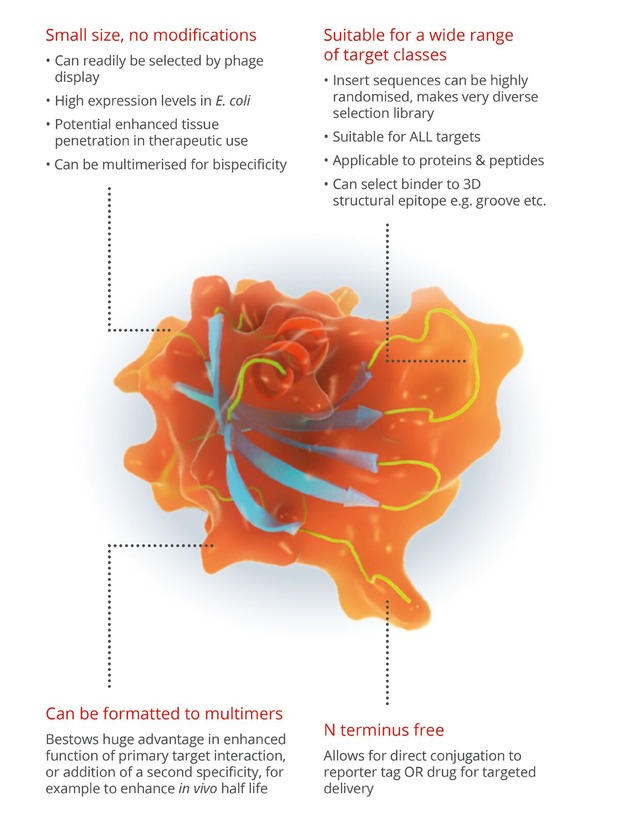
While antibodies have proven invaluable in numerous applications across biomedical research and as clinical diagnostics and therapeutics, they are not without their limitations.1, 2 Issues of cost, target specificity and reproducibility have all been raised in relation to antibody performance. Monoclonal antibodies are large and complex glycoprotein structures that are unwieldy to manufacture, and often show batch-to-batch variation.
The antibody protein structure means that sensitivity to the assay environment (such as extremes of pH, temperature or long-term storage) is highly clone specific, and finding an antibody with the desired performance characteristics can be laborious. Consequently, there has been a drive to explore recombinant antibody alternatives for performance and cost improvements across many clinical diagnostic platforms.
There has been a drive to explore recombinant antibody alternatives for performance and cost improvements across many clinical diagnostic platformsConsiderable efforts have been expended in recent years to develop non-antibody affinity reagents. An impressive number of putative antibody alternatives have resulted from this, including adnectins, affibodies, Affimers, anticalins, DARPins, fynomers, and kunitz domains. These engineered affinity reagents aim to offer increased stability to experimental conditions, the opportunity to drive target specificity through in vitro generation, and long-term security of supply, with simpler manufacture than antibodies, minimising batch-to-batch variation issues.
COPD biomarker A recent example of improved disease understanding successfully transferring to developed clinical diagnostics is the case of the chronic obstructive pulmonary disease (COPD) biomarker, soluble Receptor for Advanced Glycation End-products (sRAGE). COPD encompasses a diverse group of conditions, such as emphysema and chronic bronchitis, that impair patients’ breathing. Early mortality, high death rates and huge costs to healthcare systems all result from this rarely reversible disease. Projections for 2020 indicate COPD will be the third leading cause of death worldwide,3 making a compelling and imminent case for effective diagnostics that can identify this condition early to prevent its progression within the general population.
Validated, reliable and clinically applicable biomarkers for COPD are essential to detect the disease early in patients, identify clinically relevant patient sub-groups, and to select the most appropriate patients for clinical trials. One of the best current candidate biomarkers from a number of large-scale clinical studies is sRAGE, particularly with reference to the presence and progression of emphysema.4-8 COPD patients show lower levels of circulating sRAGE than either smoking or non-smoking individuals, and levels decrease further during periods of COPD exacerbation, correlating with both neutrophilic airway inflammation and emphysema.
[caption id="attachment_70290" align="alignleft" width="324"] Just what these affimers and what can they do?[/caption]
Just what these affimers and what can they do?[/caption]
Low levels of sRAGE are linked to COPD as it acts as a decoy receptor for pro-inflammatory ligands in the lungs. Other conditions, including diabetes mellitus, autoimmune diseases and neurological conditions have also been associated with sRAGE.9
A validated and accurate assay for sRAGE quantification is essential to allow further research into the biological function and clinical application of this biomarker. Most studies to date have used commercially available ELISA to measure sRAGE levels. However, investigations into these assays highlighted lack of sensitivity and reproducibility, and issues with calibration. A subsequent LC-MS based immunoassay strategy was established, and validated to increase the sensitivity in detection and accuracy in the quantification of sRAGE within human serum samples.10 This method made use of initial immunoprecipitation steps within a 96-well ELISA plate, for the enrichment of the biomarker protein, to increase sensitivity. Increases in assay time were potentially offset by 2-4 fold increases in sRAGE sensitivity in the LC-MS assay. Further performance analyses yielded concerns over the accuracy and batch-to-batch variability of the antibodies in this immunoassay, which were the same antibodies employed in the original ELISA assays. Consequently, the researchers explored the use of alternative reagents.
Affimer reagents, highly specific and sensitive antibody alternatives, were explored for use as affinity reagents in this potential LC-MS diagnostic assay for COPD.11 The orientation of Affimer reagents on a solid surface can be controlled for increased capture, and their high stability and robustness to the assay environment ensures their function across a wide range of assay conditions, and within a variety of potentially harsh sample types. Simple, highly reproducible manufacture of these reagents in E.coli ensures consistent batch-to-batch performance for immunoassay reagents and a completely animal-free production process.
In developing an Affimer-based LC-MS method for the quantification of sRAGE within the clinically relevant range of 0.2-10 ng/mL, a combination of two sRAGE-specific Affimer reagents were adsorbed onto microtiter plates to capture endogenous sRAGE from serum samples. Accurate quantification of sRAGE by LC-MS was shown, with both assay accuracy and precision within the acceptable limits for EMA and FDA bioanalytical assay guidelines. Further performance enhancements over the antibody-based assay were noted in the reproducibility of the Affimer reagents, and sensitivity when tested against patient samples from a variety of sources.
Liver cancer biomarker The Affimer technology has been applied in other diagnostics under development, for example a chemiluminescent immunoassay for the hepatocellular carcinoma biomarker glypican-3.12 Glypican-3 is a heparan sulphate proteoglycan, which is expressed at elevated levels in cases of hepatocellular carcinoma in human liver cancer. The reported serum levels for this biomarker differ markedly between different antibody-based diagnostic assay kits, preventing the establishment of a clear diagnostic level using this approach.
An Affimer-based immunoassay was developed by pairing Affimer reagents specific for glypican-3 with an antibody, as part of a sandwich immunoassay. This immunoassay showed wide linear range, high limit of detection, and high specificity. In comparison to commercially available glypican-3 antibody-based assay kits, the specificity offered by the Affimer-based diagnostic assay showed the closest correlation in performance to the ‘gold standard’ glypican-3 immunohistochemistry for disease diagnosis.
An interesting feature of Affimer reagents is their typical preference for non-contiguous tertiary structure targets,13, 14 rather than the continuous epitopes to which antibodies usually bind. This has enabled the development of reagents that specifically recognise complex, diagnostically relevant targets, such as a therapeutic antibody-target complex or an antibody bound to vitamin D2/D3, without binding the individual components.
This characteristic of Affimer reagents enables a range of assays to be developed that would be difficult to achieve using classical antibody-based methods. Examples of this approach include the identification of reagents to allow monitoring of drug/target complexes in pharmacokinetic or therapeutic drug monitoring assays, and conversion of a small-molecule competition assay into a sandwich assay format for improved robustness, sensitivity and dynamic range.
Overcoming antibody limitations These case studies highlight the new opportunities that the use of alternative affinity reagents can bring to the field of clinical diagnostics, to develop tests for new targets and diseases that function with increased sensitivity and improved consistency. Affimer binders offer a robust nature, high target specificity and selectivity within a variety of matrices, along with consistent performance, meeting the validation criteria for regulatory approval. Improved lot-to-lot reproducibility enabled through ease of manufacturing, combined with the rapid isolation of quality binders from recombinant phage libraries, means that these reagents can overcome some of the major limitations associated with antibodies. Namely production costs, batch variation and long developmental lead times.
As such, they represent a real alternative to traditional antibody reagents, which can be utilised to increase knowledge and availability of disease biomarkers in order to bring new diagnostics to market sooner, ultimately improving patient care. Rational design of antibody alternatives, such as Affimer reagents, will allow for the targeting of a broader swathe of molecular targets, and function across more platforms to better support the development of clinical diagnostics. While the concerns over antibody performance are increasing, the solution may already be available.
References
- Chames, P.;?Van Regenmortel, M.;?Weiss, E.;?Baty, D.?Therapeutic antibodies: successes, limitations and hopes for the future.?Br. J. Pharmacol.?2009,?157,?220–?233.
- Baker, M.?Reproducibility crisis: Blame it on the antibodies.?Nature?2015,?521,?274–?276
- Raherison, C. and Girodet, P.O. Epidemiology of COPD. Eur. Respiratory Rev. 2009 18:213-221.
- Cheng, D. T.; Kim, D. K.; Cockayne, D. A. et al. TESRA and ECLIPSE Investigators Systemic soluble receptor for advanced glycation end products is a biomarker of emphysema and associated with AGER genetic variants in patients with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2013, 188, 949-957.
- Hoonhorst, S. J.; Lo Tam Loi, A. T.; Pouwels, S. D. et al. Advanced glycation endproducts and their receptor in different body compartments in COPD.?Respir. Res.?2016,?17,?46.
- Sukkar, M. B.; Ullah, M. A.; Gan, W. J et al. RAGE: a new frontier in chronic airways disease.?Br. J. Pharmacol.?2012,?167,?1161–?1176.
- Yonchuk, J. G.; Silverman, E. K.; Bowler, R. P. et al. Circulating soluble receptor for advanced glycation end products (sRAGE) as a biomarker of emphysema and the RAGE axis in the lung.?Am. J. Respir. Crit. Care Med.2015,?192,?785–?792.
- Zemans, R. L.; Jacobson, S.; Keene, J. et al.?Multiple biomarkers predict disease severity, progression and mortality in COPD.?Respir. Res.?2017,?18,?117.
- Vazzana, N.;?Santilli, F.;?Cuccurullo, C.;?Davi, G.?Soluble forms of RAGE in internal medicine.?Int. Emerg. Med.?2009,?4,?389–?401
- Klont, F.; Pouwels, S.D.; Hermans, J. et al. A fully validated liquid chromatography-mass spectrometry method for the quantification of the soluble receptor of advanced glycation end-products (sRAGE) in serum using immunopurification in a 96-well plate format. Talanta. 2018, 182, 414-421.
- Klont, F.; Hadderingh, M.; Horvatovich, P. et al. Affimers as an Alternative to Antibodies in an Affinity LC–MS Assay for Quantification of the Soluble Receptor of Advanced Glycation End-Products (sRAGE) in Human Serum. J. Proteome Res. 2018, 17(8), 2892-2899.
- Xie, C.; Tiede, C.; Zhang, X. et al. Development of an Affimer-antibody combined immunological diagnosis kit for glypican-3. Sci. Reports. 2017, 7, 9608.
- Michel, M.A.; Swatek, K.N.; Hospenthal, M.K. et al. Ubiquitin Linkage-Specific Affimers Reveal Insights into K6-Linked Ubiquitin Signaling. Mol. Cell 2017, 68(1), 233-246.
- Robinson, J.I.; Baxter, E.W.; Owen, R.L. et al. Affimer proteins inhibit immune complex binding to Fc?RIIIa with high specificity through competitive and allosteric modes of action. Proc Natl Acad Sci USA. 2018, 115(1), E72-E81.
 Dr Robert Ford is Senior Scientist at Avacta
Dr Robert Ford is Senior Scientist at Avacta
 Ryan Hannan is a scientist at Avacta
Ryan Hannan is a scientist at Avacta