The power of partnership takes on HIV
28 Apr 2015 by Evoluted New Media
Having already made notable contributions to our understanding of how HIV works, Professor Andrew Lever talks about how his team’s most recent research is being driven forward through open collaboration with GSK
Having already made notable contributions to our understanding of how HIV works, Professor Andrew Lever talks about how his team’s most recent research is being driven forward through open collaboration with GSK
Current estimates suggest that approximately 35 million people around the world are living with human immunodeficiency virus (HIV). Of these, 1.5 million people died during 2013 alone as a result of acquired immunodeficiency syndrome (AIDS)-related illnesses1. While there are now a large number of drugs available and a range of new therapeutics in development to combat HIV, the genome of the virus is inherently prone to mutation, which can lead to the generation of drug-resistant strains. However, resistance is not a one-dimensional problem. For example, difficulties can also arise when patients fail to adhere to, or complete, anti-retroviral drug regimens, resulting in a higher frequency of these drug-resistant strains in newly acquired infections. Factors such as these have enabled the virus to acquire resistance from different sources over time, something that is driving a continuing need to develop new medicines to combat this disease.
Following initial infection with HIV, the virus begins to utilise the host’s own cellular machinery to generate additional copies of itself. These copies spread throughout the body, specifically targeting cells of the immune system, including CD4+ T-cells, macrophages and dendritic cells. As HIV is a retrovirus, it uses the enzyme reverse transcriptase to create a double stranded DNA copy from its single stranded RNA genome. The new double stranded copy is then spliced into the host’s DNA. The HIV genome can lie dormant in the nucleus of the host cell for many years, before it is stimulated to awaken and begin creating new copies of the viral RNA genome, and thus the production of new viral particles. This is an elegant process whereby the virus uses the host’s own cellular machinery to make more copies of itself, while escaping detection from the very cell type that would usually mediate an immune response.
Reverse transcription is an exceedingly error-prone process and can lead to the introduction of mutations into the viral genome every time it is duplicated and spliced into the host’s DNA. However, rather than being an Achilles heel, this is actually a key source of HIV’s ability to acquire resistance to certain drugs, as every error event results in the creation of a slightly different form of the virus. Such variation is the driving force of selection, and each new copy of the virus may house a mutation that renders it immune to the effects of certain classes of drug. The high rate of viral replication means that there is a rapid generation of new HIV phenotypes, leaving many drug developers struggling to keep up; the more we use a given drug, the more selective pressure we put on HIV to mutate and escape from a drug’s mode of action.
The primary goals for those of us working in HIV research and drug discovery are twofold. Firstly, we are keen to prolong the effectiveness of current classes of drug by overcoming resistance as it emerges. Secondly, we are working to develop new classes of drug that target underexplored areas of the HIV life cycle.
The efforts of researchers around the globe have led to the development of numerous drugs against HIV, each of which targets different stages of the virus life cycle (Figure 1). However, the majority of these have primarily focussed upon the early stages of virus infection, such as entry, reverse transcription and integration.
- Entry inhibitors target the process of virus entry into a host cell.
- Nucleoside, nucleotide and non-nucleoside reverse transcriptase inhibitors target the viral enzyme reverse transcriptase that generates the double stranded DNA copy from the original viral RNA.
- Integrase inhibitors block the viral enzyme integrase, which normally splices the viral DNA copy into the host genome
[caption id="attachment_42607" align="aligncenter" width="450"]
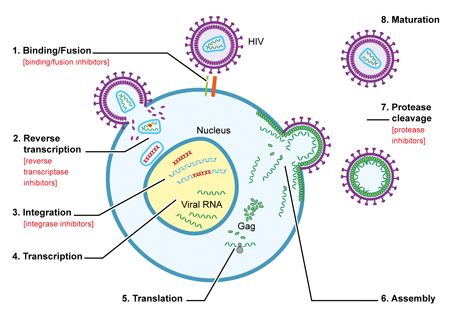 Figure 1. The life cycle of HIV and the targets of existing anti-retroviral drug classes. Most drugs currently target the early stages of the virus life cycle such as reverse transcription of the virus (drug classes indicated in red).[/caption]
Figure 1. The life cycle of HIV and the targets of existing anti-retroviral drug classes. Most drugs currently target the early stages of the virus life cycle such as reverse transcription of the virus (drug classes indicated in red).[/caption]
While informative, this list should not be considered exhaustive. For example, one class of drugs that does target a much later stage are the protease inhibitors, which block cleavage of key viral proteins that are essential to HIV progressing to a mature, infectious form. These factors operate right at the end of the virus life cycle, inhibiting the release of a new infectious particle.
In between these two ends of the life cycle there are several unexplored stages that may hold valuable insight into potential therapeutic targets. The research conducted in my own lab has focussed on building a better understanding of these aspects of the HIV life cycle. We have been able to identify a small group of compounds that exhibit promising qualities in reducing the ability of HIV to proliferate and generate additional infectious virus particles. This is an exciting new area of study that we are currently actively pursuing.
Having generated this pilot data, we were keen to expand our research in the search for new compounds of interest. While the public libraries we initially screened were invaluable in our early efforts, we were limited to less than 3000 molecules in total, so it quickly became evident that we needed to widen the scope of our programme to increase the chances of finding viable compounds for further study. When exploring the options available to academic scientists working in drug discovery, we were introduced to GlaxoSmithKline’s (GSK) new Discovery Fast Track (DFT) challenge, which provides academic scientists such as ourselves with the opportunity to work in partnership with GSK. The DFT challenge invites academics to submit a one-page abstract detailing how their research could be combined with GSK’s screening infrastructure, experience and expertise to drive drug discovery forward in an important disease area.
Having conducted the vital primary research ourselves, we were able to approach GSK with our idea for a novel way of targeting HIV. Via a very collaborative process, we worked with the DFT team to assess and discuss the suitability of our project, which culminated in our team being offered a place in the challenge programme.
In the past, we had made use of relatively low throughput assays to test our small compound libraries. While these were useful during our proof-of-concept studies, we needed support with scaling up the process in order to achieve industrial-level efficiency. Through this collaboration, we will gain access to not only the vast screening library and drug discovery expertise that GSK has to offer, but also to high throughput screening technologies and GSK’s ability to conduct research on a scale far beyond the capabilities of an academic department.
We also needed to consider what would happen should we identify any compounds of interest. For example, there are the complex regulatory aspects that we would need to navigate should our studies prove successful. This is not an area where academics tend to have expertise; our value is in our cutting-edge understanding of the biology behind the disease and our ability to come up with creative ways to explore and test new and interesting hypotheses. By partnering with GSK, we gain access to the team’s substantial amount of experience nurturing drug discovery programmes through early screening and beyond.
To be able to progress this research, we knew that working with someone like GSK was the next logical step, and my experience with the DFT program has thus far been an interesting and rewarding one. We are very hopeful that this collaboration will lead to the development of a highly targeted drug, and perhaps even an entirely new class of anti-HIV compounds. If our partnership is successful, it could have significant benefits for those living with HIV around the world, which is something everyone involved with the project is very passionate about achieving.
References:
- UNAIDS. World AIDS Day Report 2014. (2014).
The author:
Andrew M.L. Lever is Professor of Infectious Diseases and Honorary Consultant Physician at Addenbrooke's Hospital, Cambridge, UK.

