Beating the diamond-standard
4 Jan 2019 by Evoluted New Media
Forget diamond – when considering super-hard materials, says Metin Örnek, the search for an alternative is on…
How hard is super-hard? With advances in material science and engineering, comes the possibility to synthesise materials that are harder, tougher, and essentially better suited to their applications.
A super-hard material is classified as having a hardness value greater than 40 gigapascals (GPa) when measured by the Vickers hardness (Hv) test. To put that in perspective, diamond, the hardest material known to man, has a hardness value of at least 65 GPa, ranging up to 100 GPa and sometimes even higher.
Being a super hard material, diamond has found applications in a variety of areas from cutting and grinding tools to wear-resistant and protective components/coatings, and even semiconductors. However, despite being the hardest material known, diamond can deteriorate when used in the machining of ferrous metals and their alloys. It also has poor thermal stability and can oxidise at high temperatures (over approximately 760°C).
The search for an alternative, super-hard material is on.
Boron nitride (BN) is an interesting ceramic. It exists in a number of crystalline forms, or polymorphs, that offer different benefits depending on the application. Low density phases include hexagonal, rhombohedral, turbostratic, explosion, and amorphous structures, while high density phases include cubic and wurtzite structures.1
Despite being the hardest material known, diamond can deteriorate when used in the machining of ferrous metals and their alloysCubic boron nitride (c-BN) and wurtzite boron nitride (w-BN) are the two phases that offer beneficial properties including super-hardness, high thermal conductivity, and high wear resistance. These enhanced characteristics are due to diamond-like covalent bonds within the c-BN and w-BN structures.2,3 Looking at hardness specifically, testing shows c-BN to have a Vickers hardness of 45-85 GPa and w-BN to measure 35-40 GPa. Importantly, the chemical stability of c-BN and w-BN against ferrous metals and their alloys is also much better than diamond, making super-hard boron nitride the material of choice for engineering hardened or alloyed steel.
High pressure high temperature synthesis
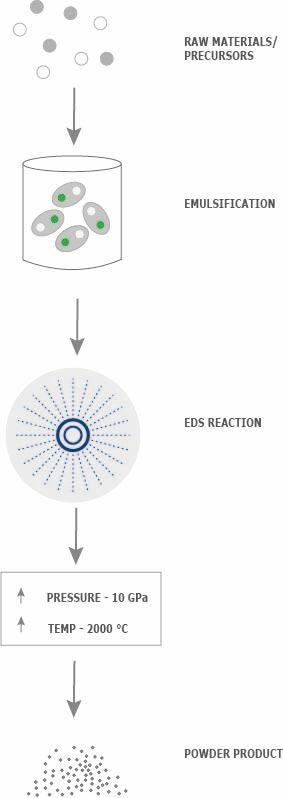
Typically, c-BN and w-BN are synthesised through high pressure high temperature (HPHT) processes. Pressures of 5-50 GPa and temperatures of 1200-2000°C are regularly used through techniques such as static and dynamic shock compressions and detonation. The extreme pressures and temperatures needed for these methods, however, make synthesis a challenge, which limits production capacity, hindering the more widespread application of c-BN and w-BN.[caption id="attachment_69345" align="alignleft" width="283"] Figure 1: Emulsion Detonation Synthesis (EDS) is a unique, proprietary method for the industrial-scale manufacture of complex, nanostructured ceramic powders.[/caption]
Figure 1: Emulsion Detonation Synthesis (EDS) is a unique, proprietary method for the industrial-scale manufacture of complex, nanostructured ceramic powders.[/caption]
The catalyst solvent method is currently the most common technique used in the industry to synthesise high-density boron nitrides. It uses an alkaline earth nitride catalyst to reduce the threshold pressure and temperature of the process but isn’t without compromises as post-processing steps are then required to remove catalyst residues. In addition, the mass production of high-density boron nitrides is still challenging using this method as it employs the conventional HPHT apparatus, which comes with production capacity limitations.
It’s clear that there is a need for a novel approach for the synthesis of high-density, super-hard boron nitride at reduced pressures and temperatures.
Recently, Rutgers University and Innovnano – a manufacturer of advanced ceramic powders – embarked on a joint project to develop a novel, scalable method for the synthesis of super-hard boron nitride. The research project applied Emulsion Detonation Synthesis (EDS)4-6 to the synthesis of boron nitride.
Quite a shock…
EDS is a unique, proprietary method for industrial-scale manufacture of complex, nanostructured ceramic powders. A leading shock wave (termed the detonation wave) is formed during the detonation process, reaching high pressures of up to 10 GPa and inducing a chemical reaction. During the reaction, very high temperatures of up to 2000ºC are obtained followed by a fast cooling rate of 109 K/s. All of these features, combined with its flexibility in terms of emulsion formulations, provide a favourable environment for the production of super-hard boron nitrides with nanometre-sized grains and enhanced homogeneity on a large scale.
In terms of tailor-made synthesis, EDS allows excellent flexibility in the possible precursors used. This permits greater manufacturing control over the purity, chemical composition, structure, and final properties of the synthesised powder. In this way the process enables the manufacture of powders with properties that can be carefully designed to suit application requirements. It’s also possible to control the detonation atmosphere by designing the composition of the emulsion mixture. For example, introducing nitrogen-rich compounds and/or oil emulsions to prevent oxidation during the reaction.
The team applied the EDS process to boron nitrides for the first time using a low density hexagonal form (h-BN) as the precursor. Initially, the team were able to demonstrate the production of a metastable explosion form (e-BN) grains embedded in h-BN matrix.7 This clearly demonstrated the potential of EDS for creating new metastable phases.
However, e-BN is still a low-density phase of boron nitride that does not exhibit the desired super-hard characteristics and so the we carried out further investigations. For example, we looked at the effects of precursor particle size, and the level of detonation wave pressure.
As a result of these investigations we recently observed the transformation from low density h-BN to super-hard w-BN during the EDS reaction.8 The same h-BN precursors were used but higher pressers were generated through a high shock transmission from the detonation wave, increasing the emulsion density. EDS has the ability to achieve a homogeneous dispersion of starting precursors that is transformed into an emulsion formulation, as well as a better impedance matching between emulsion and h-BN particles. With these abilities, EDS offers a synthesis solution for the production of super-hard metastable boron nitride.
What next?
Currently, the w-BN yields obtained through EDS are relatively low, and so this is the next focus for research. With EDS, the degree of transformation to super-hard boron nitride is closely related to the process parameters, including pressure, particle size of the starting material, and the cooling rate.Higher pressures are expected to increase the yield of the super-hard phases, while the use of starting materials with particles that are too large can lead to limited or no transformation to super-hard boron nitride.9 A faster cooling rate also results in increased transformation.10 Further investigation is being performed to optimise these process parameters and maximise potential w-BN yield with EDS.
The approach described here utilises the unique environment created during EDS – high temperatures, high pressures, and fast cooling rates to create super-hard boron nitride from BN-containing precursors. In addition to this, researchers are also working on a new approach that uses boron- and nitrogen-containing raw materials such as melamine and boric acid. Unlike in the process we have outlined here, this means that no forms of boron nitride itself are needed from the outset. Instead, super-hard c-BN and w-BN are expected to be synthesised from direct reactions in a one-step synthesis process. This eliminates a synthesis step, resulting in significant cost savings and increased production efficiency.
With EDS, super-hard just got easier. Taking advantage of the unique environment created as a result of the detonation wave, the production of metastable boron nitride with super-hard characteristics is now possible. The potential of this technology is great. With further work on yield and boosts in manufacturing efficiency, it could be possible to produce industrial quantities of super-hard materials in the very near future, and with applications across numerous industries, this promises to be a great advantage.
References
- R. Haubner, M. Wilhelm, R. Weissenbacher, B. Lux, Boron Nitrides — Properties, Synthesis and Applications, in: M. Jansen (Ed.) High Performance Non-Oxide Ceramics II, Springer Berlin Heidelberg (2002), pp. 1-45.
- C. Ji, V.I. Levitas, H. Zhu, J. Chaudhuri, A. Marathe, Y. Ma, Shear-induced phase transition of nanocrystalline hexagonal boron nitride to wurtzitic structure at room temperature and lower pressure, Proceedings of the National Academy of Sciences, 109 (2012) 19108-19112.
- L. Vel, G. Demazeau, J. Etourneau, Cubic boron nitride: synthesis, physicochemical properties and applications, Materials Science and Engineering: B, 10 (1991) 149-164.
- E.M.D.S. Antunes, J.M.C. Da Silva, A.L.C. Lagoa, Process for nanomaterial synthesis from the preparation and detonation of an emulsion, products and emulsions thereof, US Patent 9,115,001, 2015.
- N. Neves, A. Lagoa, J. Calado, A.B. do Rego, E. Fortunato, R. Martins, I. Ferreira, Al-doped ZnO nanostructured powders by emulsion detonation synthesis–Improving materials for high quality sputtering targets manufacturing, Journal of the European Ceramic Society, 34 (2014) 2325-2338.
- S.M.P. Da Silva, J.M.C. Da Silva, Continuous process for nanomaterial synthesis from simultaneous emulsification and detonation of an emulsion, US Patent 9,327,257, 2016.
- M. Örnek, K.M. Reddy, C. Hwang, V. Domnich, A. Burgess, S. Pratas, J. Calado, K.Y. Xie, S.L. Miller, K.J. Hemker, Observations of explosion phase boron nitride formed by emulsion detonation synthesis, Scripta Materialia, 145 (2018) 126-130.
- M. Örnek, C. Hwang, K.Y. Xie, S. Pratas, J. Calado, A. Burgess, V. Domnich, K.J. Hemker, R.A. Haber, Formation of metastable wurtzite phase boron nitride by emulsion detonation synthesis, Journal of the American Ceramic Society, 101 (2018) 3276-3281.
- R.C. Ripley, F. Zhang, F.S. Lien, Acceleration and heating of metal particles in condensed matter detonation, Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences, 468 (2012) 1564-1590.
- A. Aleksenskii, M. Baidakova, A.Y. Vul, V.Y. Davydov, Y.A. Pevtsova, Diamond-graphite phase transition in ultradisperse-diamond clusters, Physics of the Solid State, 39 (1997) 1007-1015.
 Dr Metin Örnek is a post-doctoral research associate working on characterization of energetic materials and dynamic mechanical behaviour of materials at Purdue University
Dr Metin Örnek is a post-doctoral research associate working on characterization of energetic materials and dynamic mechanical behaviour of materials at Purdue University