Making a value judgement
7 May 2019 by Evoluted New Media
Bioethics has become a global endeavour – and a good thing too says Catherine Joynson. As we push the boundaries of what we do with biology, we need to ask ‘should we’ as well as ‘can we’…
All good scientists will be aware that sometimes, perhaps even often, their research will have implications for people and wider society beyond the laboratory. Considering those implications – the potential benefits and harms – and how they fit with the values people hold important, is how bioethicists spend their days.
Bioethics has become a global occupation, with large amounts of academic research being undertaken across the world and government-appointed bioethics committees now in existence in many countries.
New discoveries and technologies constantly challenge us and can make past dilemmas seem simple in comparison
The UK is no different, and we have a thriving bioethics community. Something that sets us apart, however, is the absence of a state-run bioethics committee. There is some history behind this. In the late 1980s, the quest to map the entire human genome was gaining momentum, and the lack of a UK national bioethics committee to consider the ethical issues this raised was emerging as a problem. Parliamentary debates on abortion and embryo research took place without, according to the British Medical Journal, “a clear and authoritative source of professional advice or a collective lead on the wider ethical and policy issues”.1 Yet the government of the day were not minded to set up a national bioethics committee.
So, in 1991, the Nuffield Foundation established the Nuffield Council on Bioethics, an independent body tasked with clarifying the ethical issues raised by biological and medical research. Wellcome and the Medical Research Council joined the Nuffield Foundation as co-funders of the Council in 1993, and it has remained that way ever since.
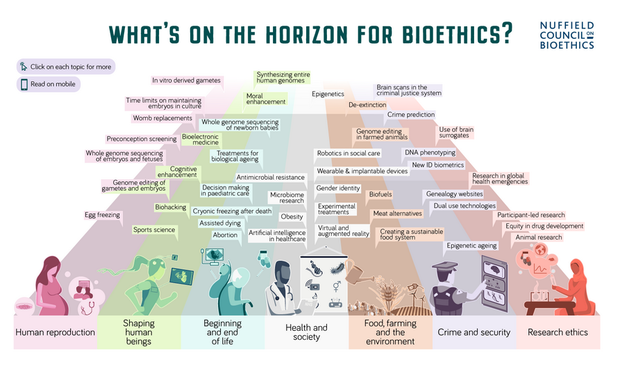
[caption id="attachment_92101" align="alignleft" width="358"]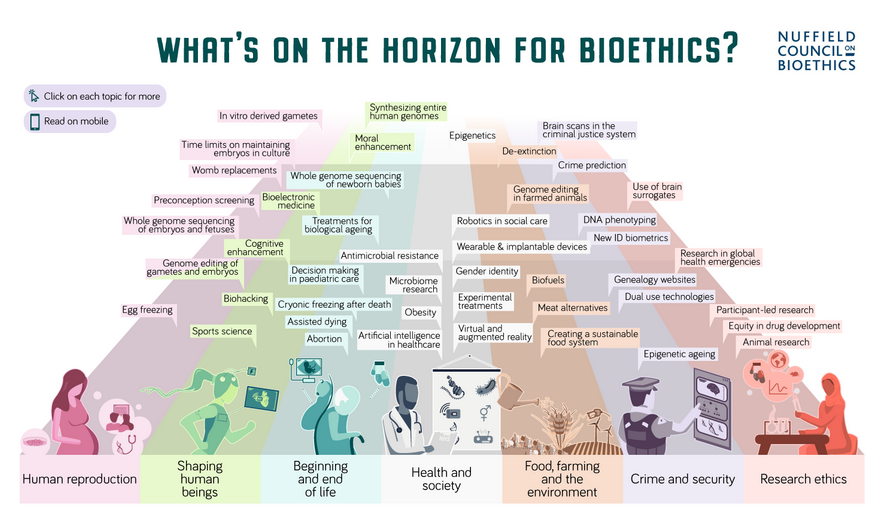 Developments that the Nuffield Council on Bioethics have in their sights[/caption]
Developments that the Nuffield Council on Bioethics have in their sights[/caption]
At the Nuffield Council, we see ourselves as occupying a space between academia, industry and society on the one hand, and the world of policy making on the other. We have no political, religious, financial or other affiliations, yet we engage with all perspectives. We aim to offer pragmatic policy advice on complex bioethics problems and are not afraid to challenge the status quo. When we asked whether it might be ethical to compensate organ donors in order to increase organ donation rates, the Guardian accurately said “The Nuffield Council on Bioethics never shrinks from the unthinkable.”2
Genomethics Genomics has long been an important focus for bioethicists. Most recently, genome editing techniques such as CRISPR-Cas9 have received global attention. In November 2018, it emerged that gene-edited twin girls had been born to a Chinese couple. The editing, it was claimed, had targeted the CCR5 gene, with the aim of introducing immunity to HIV and some other viruses. Shortly before this development, the Nuffield Council published the findings of a two-year inquiry on the use of genome editing in human reproduction. We examined the use of genome editing in the context of assisted reproduction to alter a DNA sequence of an embryo, or of a sperm or egg cell prior to fertilisation, with the aim of influencing the inherited characteristics of the resulting person, and future generations.
After extensive consultation and deliberation, we concluded that the use of heritable genome editing interventions is not, in principle, morally unacceptable, so there is no reason to rule it out altogether. However, the circumstances under which its use could be morally permissible – including that there has been a sufficient opportunity for broad public debate, research has been done to establish standards of safety, and risks of adverse effects for individuals, groups and society as a whole have been assessed and mitigated against – do not exist at present, anywhere. We must therefore get appropriate measures in place now to secure a responsible path to innovation and, perhaps, uptake of this technology.3
We did not expect our findings to be applied to real-world events so soon. We strongly condemned the actions of the Chinese scientist responsible for editing of the genomes of the babies in China. My Nuffield Council colleague Peter Mills said “...it is a premature, inexplicable and possibly reckless intervention that may threaten the responsible development of future applications of genome editing.” Bioethicists around the world, including those in China, concurred that the actions of the scientist involved were unethical. The Chinese Government later said that the scientist acted “illegally and in pursuit of fame and fortune”,4 and it recently published draft regulations that require national approval for clinical research involving gene editing and other high-risk biomedical technologies.5 I wonder whether such expedient action would have been taken at a time before bioethicists had the powerful global voice that they do today.
Future ethical worries We are always on the lookout for the latest developments in science to ensure we are anticipating bioethical issues that might arise in future. Listening to scientists themselves is vital, and it seems these issues cut across all areas of scientific research. We recently set out the developments that are currently in our sights in an interactive infographic. It covers seven themes: human reproduction; shaping human beings; beginning and end of life; health and society; food, farming and the environment; crime and security; and research ethics.
One area we are actively pursuing at the moment is the increasing use of implantable medical devices. These devices include those that replace body parts such as hip implants, deliver medication such as insulin, and provide support to organs and tissues such as pacemakers. New medical devices emerge at a much higher rate than new medicines, and about 400,000 devices have been approved for use in the EU. Medical devices can improve quality of life for patients and can be lifesaving. However, regulation of medical devices has been criticised for being too lenient and lacking transparency.6
Some devices have caused serious side-effects after being used in large numbers of patients without, it is claimed, adequate testing. For example, hundreds of women who were fitted with a vaginal mesh implant to treat pelvic organ prolapse have been left with life changing injuries and complications. The European-wide system of regulation is meant to protect patients from this kind of harm, but it also aims to support innovation and growth. In this case, it might be that the latter took priority over the former.
As well as addressing concerns about safety, any changes to regulation in this area will need to account for the fact that devices are increasingly connected to other devices, or to other networks. The ‘internet of things’ expands possibilities for data gathering, monitoring and analysis, and for upgrading medical devices after implantation. But this might also make devices more vulnerable to error and attack. Famously, the former US Vice President Dick Cheney had his pacemaker’s wifi connection disabled to thwart possible assassination attempts.
It’ll only get trickier A further factor to consider is the strong desire by patients to access innovative treatments, particularly if they are in a desperate situation or have exhausted all other options. Patients can be willing to take higher risks than usual if there is a possibility, even very slight, that a treatment could be effective for them. In a recent publication on patient access to experimental treatments, we highlighted the important responsibilities of healthcare professionals to support patients to make informed decisions about treatments for which evidence is limited.7
Sometimes people ask us, after almost 30 years in the business, whether there are any truly new bioethics issues left for us to consider. We tell them that new discoveries and technologies constantly challenge us and can make past dilemmas seem simple in comparison. We are seeing the convergence of different advancements in research and technology to create novel questions. For example, in the February issue of this magazine, Axel Schumacher highlighted the potential of blockchain technology to be used by individuals in a highly controlled way to share, or even sell, their genome to others in light of the availability of cheaper and more advanced genome sequencing technology. The idea of a monetised DNA marketplace, managed by individuals themselves, turns previous debates about consent processes and breaches of privacy on their heads. The increasing use of artificial intelligence (AI) technologies in healthcare and research also raises new questions about, for example, the potential for AI to make erroneous decisions, the question of who is responsible when AI is used to support decision-making, and difficulties in validating the outputs of AI systems.8
In addition, these developments are occurring within changing social contexts. There is an increasing focus on the potential for science to drive economic growth, a rising influence of social media and online activity, variable levels of trust in institutions, and shifts in our understanding and expectations of areas such as data privacy.
Being responsive to these changes, and keeping abreast of the very latest scientific developments, will be keeping the bioethics community busy for some time to come.
Author:
 Catherine Joynson is Assistant Director at the Nuffield Council on Bioethics
Catherine Joynson is Assistant Director at the Nuffield Council on Bioethics
References:
1 Lock S (1990) Editorial: Towards a national bioethics committee BMJ 300: 1149-1150.
2 Guardian (21 April 2010) Organ transplants: Relative ethics. https://www.theguardian.com/commentisfree/2010/apr/21/organ-donation-nuffield-council-bioethics
3 Nuffield Council on Bioethics (2018) Genome editing and human reproduction: social and ethical issues http://nuffieldbioethics.org/project/genome-editing-human-reproduction
4 BBC (21 January 2019) He Jiankui: China condemns 'baby gene editing' scientist https://www.bbc.co.uk/news/world-asia-46943593
5 Science (28 Feb 2019) China tightens its regulation of some human gene editing, labeling it ‘high-risk’ https://www.sciencemag.org/news/2019/02/china-tightens-its-regulation-some-human-gene-editing-labeling-it-high-risk
6 BBC News (25 Nov 2018) Patients given unsafe medical implants https://www.bbc.co.uk/news/health-46318445
7 Nuffield Council on Bioethics (2018) Bioethics briefing note: Patient access to experimental treatments http://nuffieldbioethics.org/wp-content/uploads/Patient-access-to-experimental-treatments.pdf
8 Nuffield Council on Bioethics (2018) Bioethics briefing note: Artificial intelligence (AI) in healthcare and research http://nuffieldbioethics.org/wp-content/uploads/Artificial-Intelligence-AI-in-healthcare-and-research.pdf